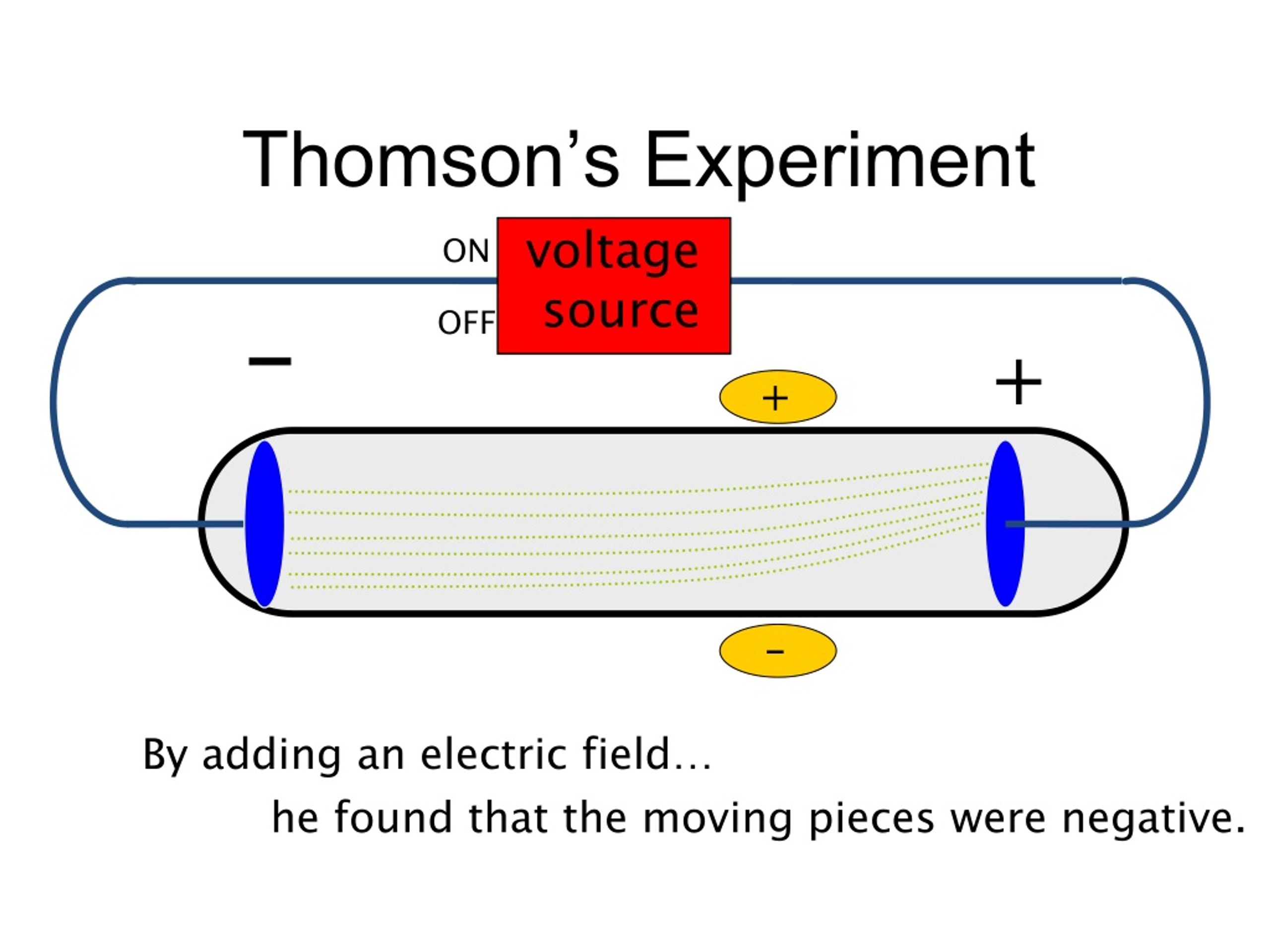
Thomson invented to test the theory that negative charges in an atom were real. Thomson is the scientist that discovered electrons through an experiment called the Cathode Ray Experiment. 
What did the cathode ray experiment discover quizlet? Particles of the cathode rays are fundamental to all matter. Matter contains positive and negative charge. What were the four things that were validated by his cathode ray experiments? – Cathode rays have mass. What was validated by the cathode ray experiment? … Thomson showed that cathode rays were composed of a previously unknown negatively charged particle, which was later named the electron. … Rutherford’s gold foil experimentĬathode rays (electron beam or e-beam) are streams of electrons observed in discharge tubes. Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. … Rutherford’s gold foil experiment showed that the atom is mostly empty space with a tiny, dense, positively-charged nucleus. What Did The Cathode Ray Experiment Prove? 38 Cathode Ray Tube Experiment and Charge To Mass Ratio of an Electron.36 Cathode Ray Tube | Definition | Charatersitics | Diagram.35 Discovery of the Electron: Cathode Ray Tube Experiment.34 What evidence from the cathode ray tube experiment support the claim that electrons have a negative charge quizlet?.33 What evidence from the cathode ray tube experiment support the claim that electrons have a negative charge?. 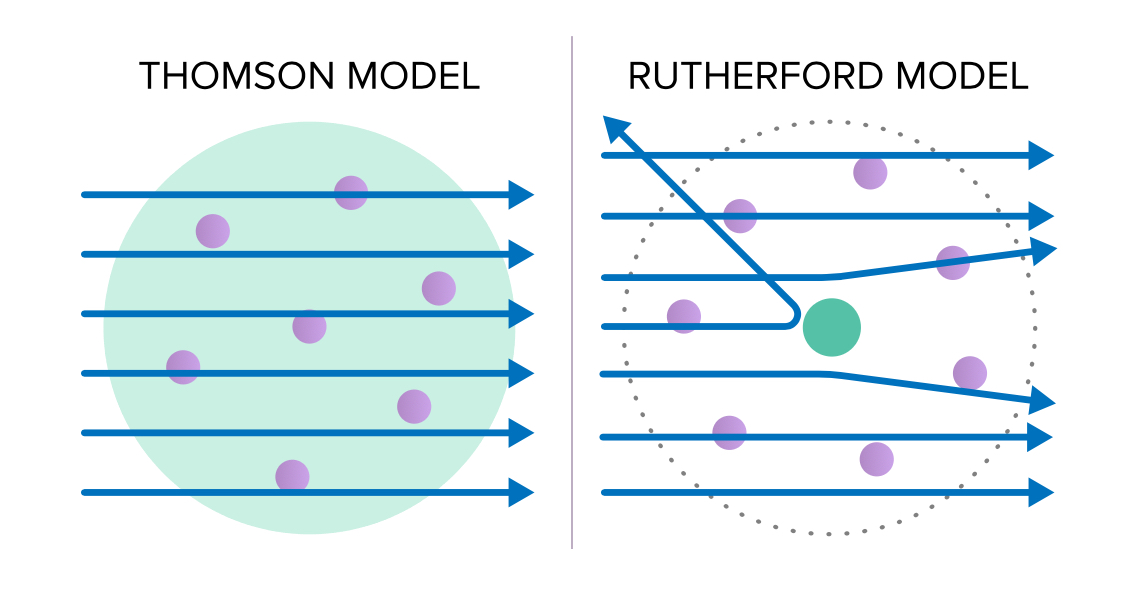
32 How did Thomson determine that the cathode ray particles were a new particle and not simply an atom molecule or ion carrying a negative charge?.30 What are the properties of cathode rays?.29 How are cathode rays produced what are the five major characteristics?.28 Which piece of evidence proves that atoms are neutral?.27 What experiment proved atoms contain positive negative and neutral particles?.26 What are observations of cathode ray tube experiment What are the important conclusions drawn from above observations?.25 How did Rutherford’s experimental evidence led to the development of a new atomic model?.24 Which experiment led to the discovery of electron and how?.23 How did experimental evidence support the development of atomic theory?.22 Is cathode ray an electromagnetic wave?.21 Why are cathode rays produced at low pressure?. 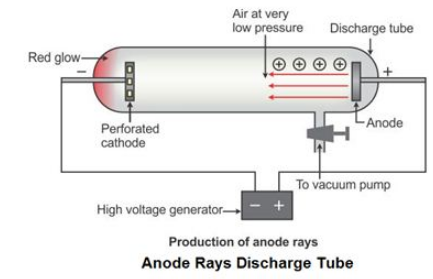
20 Why do cathode rays produce fluorescence?.19 What characteristic of cathode rays led scientists to believe that they were negatively charged?.18 What was the evidence that the cathode rays were particles with charge and mass?.17 How did the cathode ray tube show evidence of electrons?.16 How did Thomson’s experiments prove the existence of electrons?.15 What did the oil drop experiment prove?.13 What did Rutherford think was going to happen?.12 What two pieces of knowledge did Thomson learn from his cathode ray tube experiment about atomic structure?.11 What did JJ Thomson’s cathode ray experiments determine quizlet?.10 Are cathode rays fundamental to all matter?.9 How did the cathode ray tube experiment lead to the conclusion that atoms contain electrons?.8 What experimental evidence led to the development of this atomic model from the one before it?.7 Which gas is used in cathode ray experiment?.6 Which scientist discovered the electron using his cathode ray experiment?.4 What did the cathode ray experiment discover quizlet?.3 What was validated by the cathode ray experiment?.2 What does study of cathode rays show?.1 What Did The Cathode Ray Experiment Prove?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
